Step 1 – Identification of Potential Hazards
- All relevant clinical and non-clinical data are reviewed.
- Hazard classification and labeling are reported.
Step 2 – Identification of “Critical Effects”
Preferably from reported clinical or workers exposure studies.
Critical effects in humans from mechanistic studies and pharmacodynamics data are discussed in the report.
Toxicological effects can be
- Skin or Respiratory Irritation
but also
- Sensitisation,
- Acute and chronic systemic effects,
- Genotoxicity,
- Carcinogenicity and
- Reproductive toxicity effects.
Step 3 – Establishing a Point of Departure for OEL Calculation
A No Observed Adverse Effect Level ("NOAEL") corresponding to the highest dose tested at which no “critical” effect occurs should be determined for each critical effect identified.
- When several NOAELs are available,
=> the lowest value and the most sensitive animal are selected.
- In the absence of NOAEL,
=> a Lowest Observed Adverse Effect Level ("LOAEL") in adults can be used.
First-in-human-Phase 1 clinical studies are reviewed in order to identify a human NOEL.
NOEL in healthy volunteers is the highest exposure level at which there are no effects (adverse or non-adverse) and is favored to calculate an OEL over NOAEL in animals.
Step 4 – Selection of several correction factors
Several adjustments factors are used to account for various uncertainties:
- Guidance on Information Requirements and Chemical Safety Assessment IR&CSA Guidance
- Guidance on Assessment Factors to Derive a DNEL ECETOC Technical Report No 110
The specific pharmacokinetics of each drug product are taken into consideration to adjust the bioavailability factor.
Step 5 – OEL calculation
The derivation of occupational exposure limit is based on the equation published by
- Sargent and Kirk (Am Ind Hyg Assoc J. 1988;49(6):309-313),
- Galer et al. (Regul Toxicol Pharmacol. 1992;15(3):291-306),
- Schwartz (Hum Ecol Risk Assess: Int J. 1995;1(5):527-543) and
- Naumann and Weideman (Hum Ecol Risk Assess: Int J. 1995;1(5):590-613)
based upon:
- from a NOAEL/LOAEL,
- a standard worker body weight (70 kg),
- safety factors and
- human breathing rate (typically assumed to be 10 m3/8 h workday).
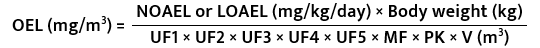
Step 6 − OEB determination
Two Occupational Exposure Banding categorizations are proposed using tables of 5 or 4 categories, presented
- in the Prevention Technical Note NTP 1.104 INSST and
- in Occupational health categorization and compound handling practice systems—roots, application and future (Chem Health Safety. 2005;12(4):20-26) which outlines the SafeBridge method.
These tables require the use of all the toxicological data collected to determine the OEB.
All Tox by Design Occupational Exposure Limit risk assessment reports are duly validated and signed by an European Registered Toxicologist expert.
In the event you are performing this exercise for innovative compounds GMP scale up manufacturing, please be noted Tox by Design is duly accredited for the French Research Tax Credit CIR.
Feel free to contact us using below automated email request to receive a quotation for Occupational Exposure Limit for your Active Pharmaceutical Ingredient, New Chemical or Biological Entity or any Chemicals substances deployed in your GMP manufacturing lines.