The Tox by Design expert team has developed a methodology to accelerate data collection and toxicity assessment of the chemicals exposed to workers operating a pharmaceutical production facility.
This generic risk assessment applied to liquids and solids only. It does not apply to gases or to liquids used above their boiling point.
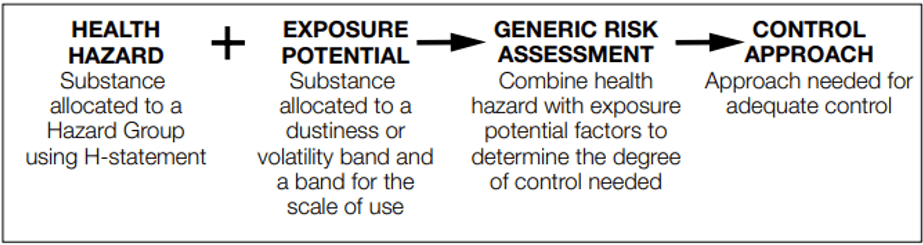
Tox by Design Chemical Occupational Exposure Risk Assessment Steps
- Step 1: Group hazards that have ‘adequate control’ at similar airborne concentrations (toxicological classification and CLP labeling)
- Step 2: Group the ‘physical properties’ with ‘amounts used’ to assign to exposure predictor bands with similar potentials for exposure.
- Step 3: Assess the anticipated exposure by applying each control approach to each exposure predictor band, based on expert judgment (each control approach gives an expected reduction in exposure).
- Step 4: Compare the predicted or anticipated exposure concentrations in air with the ‘acceptable’ airborne concentrations established in regards to hazard groups, to help select a suitable control approach.
Youu can check Tox by Design methodology for Chemical Occupational Exposure Risk Assessments, signed by an European Registered Toxicologist expert here.
In the event you are performing this exercise for innovative compounds GMP scale up manufacturing, please be noted Tox by Design is duly accredited for the French Research Tax Credit CIR.
Feel free to contact us using this email link to receive a quotation for Tox Chemical Occupational Exposure procedure used on your manufacturing line.